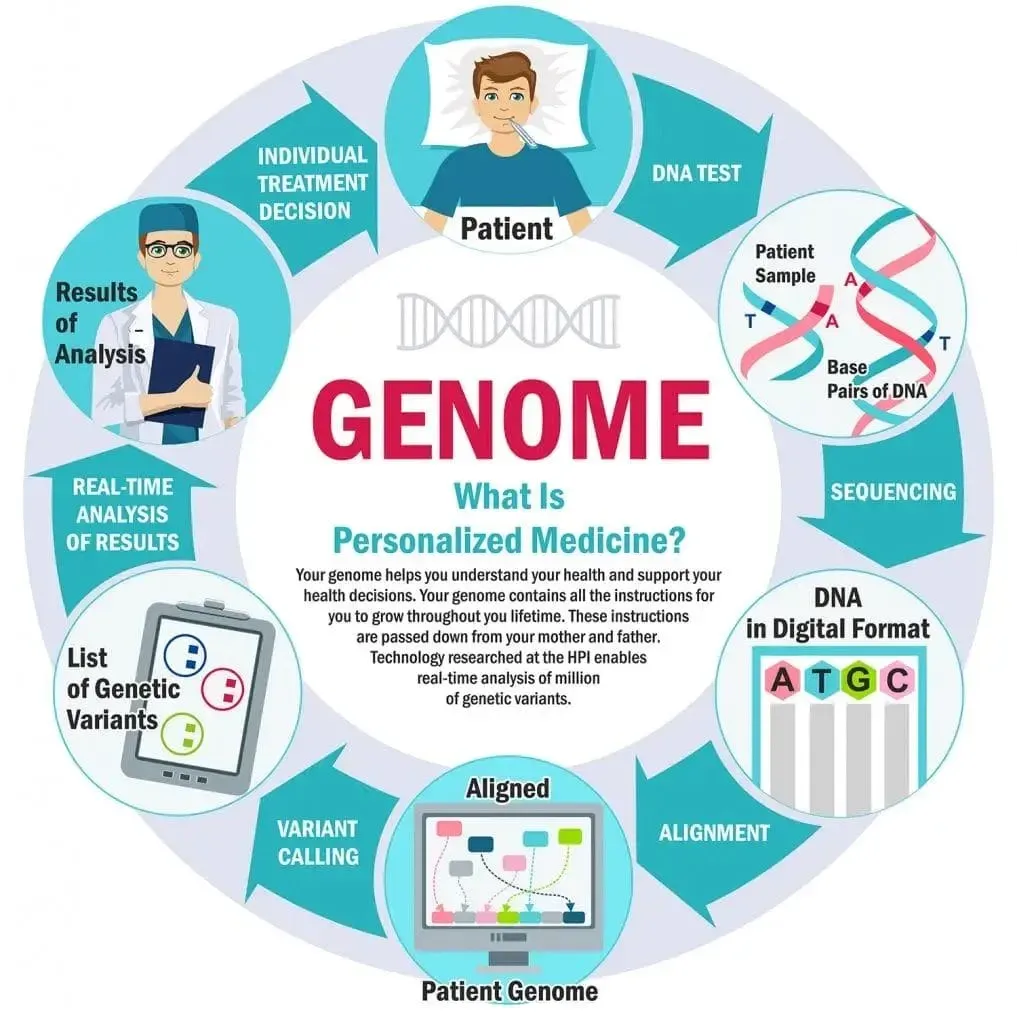
Genomics in personalized medicine is reshaping how clinicians tailor care to each patient. By integrating genomic data with clinical information, this approach advances precision medicine and guides decisions about prevention, diagnosis, and treatment. Advances in sequencing technologies are accelerating genome-informed decisions across many specialties. As data analytics improve, clinicians can interpret variants more reliably and move beyond one-size-fits-all strategies. Ultimately, patients stand to benefit from clearer options, safer therapies, and care that aligns with their unique biology.
In practical terms, this shift is often described as genome-guided or DNA-based care, where a patient’s genetic profile informs the plan at every step. Genetic profiling facilitates risk assessment, targeted testing, and more precise therapy choices—ideas that mirror pharmacogenomics and genome-informed decision support. As healthcare ecosystems adopt interoperable data platforms, clinicians can apply these insights with confidence, making care more proactive and personalized. This evolving approach also emphasizes data quality, patient consent, and collaboration across clinicians, laboratories, and families.
Genomics in personalized medicine: From concept to clinic
Genomics in personalized medicine has evolved from a theoretical concept into a practical framework that informs prevention, diagnosis, and treatment. Clinicians increasingly use an individual’s genetic information to interpret risk, guide screening strategies, and select interventions that align with a patient’s molecular profile. This genome-guided approach embodies the core goal of precision medicine: to move beyond one-size-fits-all care toward therapies that reflect unique biology and life context.
As sequencing technologies become faster and more affordable, genomic data are woven into routine practice across specialties. Pharmacogenomics, for example, helps determine how a patient metabolizes drugs, while tumor genomics reveals driver mutations that can steer targeted therapies. The result is a care pathway that is more proactive, with prevention and treatment decisions tailored to the person, not just the disease.
Genomic sequencing in healthcare: Driving targeted interventions
Genomic sequencing in healthcare—whether whole-genome, whole-exome, or targeted panels—serves as a powerful catalyst for targeted interventions. Sequencing uncovers mutations, copy-number changes, and chromosomal rearrangements that can redefine risk, prognosis, and therapeutic options. When combined with clinical data, these findings enable clinicians to chart course-correcting strategies that are more likely to yield meaningful benefit for individual patients.
The interpretation of sequencing results relies on curated databases, population genetics insights, and multi-omics integration. AI-assisted annotation helps distinguish pathogenic variants from benign variation, prioritize potential targets, and translate complex data into concrete clinical actions. Integrating genomic findings into electronic health records and decision support systems then supports day-to-day choices—shifting care from reactive treatment to tailored, genome-informed care.
Pharmacogenomics: Tailoring drug choices and dosing
Pharmacogenomics is a practical facet of precision medicine that explains why people respond differently to the same medication. Genetic differences in drug-metabolizing enzymes, transporters, and receptors influence efficacy and the risk of adverse effects, guiding safer dosing and more effective therapies. In cardiovascular care, psychiatry, and beyond, pharmacogenomic insights help clinicians select drugs and adjust doses to fit a patient’s genetic profile.
By embedding pharmacogenomics into clinical workflows, clinicians reduce trial-and-error prescribing and shorten the path to an optimal regimen. This approach embodies the promise of precision medicine: treatments that work with fewer side effects and faster therapeutic gains. As testing becomes more accessible, patients benefit from personalized plans that consider both genetics and the broader clinical picture.
Precision medicine and multi-omics: A data-driven decision framework
The promise of precision medicine grows as clinicians integrate genomic data with other types of information—electronic health records, wearable sensors, and lifestyle data. Multi-omics fusion, including genomics, transcriptomics, and proteomics, yields a richer view of biology than any single data stream alone. This integrated approach supports more nuanced risk stratification and tailoring of interventions to individual biology.
Artificial intelligence and machine learning play a central role in extracting actionable patterns from this data-rich environment. By forecasting drug responses, identifying high-risk patients, and prioritizing therapeutic options, AI accelerates decision-making without sacrificing clinical judgment. The result is a data-driven, patient-centered framework that aligns with the goals of precision medicine and improves outcomes across care settings.
Tailored therapies across oncology, cardiology, and rare diseases
In oncology, tumor sequencing informs targeted therapies, immunotherapies, and combination regimens aligned with the molecular drivers of each tumor. This tumor-centric approach illustrates how genomics supports tailored therapies that optimize response rates while minimizing collateral damage to healthy tissue. Similar strategies are increasingly applied in cardiology and neurology, where genetic insights refine drug choices and monitoring.
Beyond common cancers, sequencing can identify the genetic basis of rare diseases, enabling access to disease-specific therapies, relevant clinical trials, or supportive care options. The shared thread is a genomics-informed path to targeted interventions that prioritize the patient’s unique biology, family history, and preferences, reinforcing the shift toward personalized care across the care continuum.
Ethics, access, and the future of precision medicine
Despite rapid progress, ethical and access-related challenges remain central to the deployment of genomics in clinical care. Costs, data privacy, and consent for large-scale sequencing and data sharing require robust governance. Equity concerns demand concerted efforts to ensure diverse populations are represented in genomic databases so interpretations remain valid across groups.
Policy, reimbursement, and clinician education are critical to sustaining momentum. As genomic data become more integrated into routine care, health systems must balance innovation with practical considerations—ensuring that patients receive high-quality, genome-informed care without widening disparities. Looking ahead, the continued evolution of precision medicine holds promise for broader adoption, real-world impact, and improved health outcomes.
Frequently Asked Questions
What is Genomics in personalized medicine and why is it important?
Genomics in personalized medicine is the use of an individual’s genetic information to guide prevention, diagnosis, and therapy. This approach is a core part of precision medicine, enabling clinicians to tailor interventions to the patient’s unique genome. By leveraging sequencing data and interpretation tools, it aims to improve outcomes, reduce adverse effects, and support proactive care.
How does pharmacogenomics shape Genomics in personalized medicine and influence drug therapy?
Pharmacogenomics studies how genetic differences influence drug metabolism and response. In Genomics in personalized medicine, this science informs safer dosing and more effective therapies across conditions such as cardiovascular and psychiatric disorders. It helps implement precision medicine by moving from trial-and-error prescribing to tailored therapies based on a patient’s genetic profile.
What is the role of genomic sequencing in healthcare within Genomics in personalized medicine?
Genomic sequencing in healthcare is used to uncover mutations, copy-number changes, or gene alterations that drive disease and guide treatment choices. In Genomics in personalized medicine, tumor profiling and germline testing can identify actionable targets and inform targeted therapies. This sequencing-driven insight supports precision medicine by aligning treatment with the patient’s molecular biology.
How are tailored therapies developed from genomic data in Genomics in personalized medicine?
Tailored therapies in Genomics in personalized medicine are developed by linking genomic variations to therapeutic strategies. By identifying driver mutations in cancer or disease-causing variants in inherited conditions, clinicians select targeted therapies, immunotherapies, or clinical trial options. This approach epitomizes precision medicine, using genomic data to customize care for each patient.
What are the main challenges and ethical considerations in Genomics in personalized medicine?
Several challenges and ethical considerations accompany Genomics in personalized medicine. Costs for sequencing and data interpretation, privacy risks, and the handling of incidental findings require robust governance and consent processes. Ensuring equity by including diverse populations in genomic databases is essential to avoid biases and expand benefits. Clinicians also need workflows that support genetic testing without delaying care.
What does the future hold for patients in Genomics in personalized medicine?
The future of Genomics in personalized medicine promises deeper integration of genomic sequencing, AI-driven interpretation, and electronic health records into routine care. Patients may benefit from real-time pharmacogenomics dashboards, rapid tumor profiling, and proactive prevention guided by genetic risk. Across health systems, tailored therapies and precision medicine could become the standard for a broader range of diseases.
| Key Point | Description |
|---|---|
| Core concept | Genomics in personalized medicine uses an individual’s genetic information to guide prevention, diagnosis, and therapy, shifting from one-size-fits-all to genome-guided care with aims to improve outcomes and reduce adverse effects. |
| Sequencing technologies | Whole-genome, whole-exome, and targeted panels generate data revealing mutations and structural changes; interpretation pipelines and curated databases turn data into actionable insights. |
| Clinical actionability and pharmacogenomics | Sequencing accuracy, variant interpretation, and clinical actionability enable tailored treatment plans; pharmacogenomics informs drug choice and dosing to optimize efficacy and safety. |
| Data integration and decision support | Genomic data are integrated into electronic health records and clinical decision support to translate complex information into day-to-day care. |
| Multi-omics and AI | Combining genomics with transcriptomics, proteomics, and phenotypic data, with AI/ML, improves risk prediction and therapy prioritization. |
| Real-world impact and specialties | Oncology, cardiology, neurology, and rare diseases leverage tumor profiling, pharmacogenomics, and genetic insights to tailor therapies and reduce side effects. |
| Challenges and ethics | Cost, data interpretation, privacy, equity, consent, incidental findings, workflow integration, and reimbursement shape adoption and require governance. |
| Future outlook | As sequencing costs fall and data sharing improves, expect deeper integration, real-time pharmacogenomics, population screening, and proactive, genome-informed care. |
Summary
Genomics in personalized medicine is redefining how we prevent, diagnose, and treat disease by leveraging an individual’s genetic information to tailor care. This descriptive overview highlights how sequencing technologies, interpretation, data integration, and AI-enabled decision making come together to translate complex data into practical, patient-specific choices. Across oncology, cardiology, neurology, and rare diseases, genome-guided strategies aim to move care from reactive to proactive, reducing adverse effects and improving outcomes. As data resources grow and governance evolves, Genomics in personalized medicine holds promise for smarter prevention, targeted therapies, and healthier populations.